
Non-Opioid Treatment for Painful Diabetic Neuropathy
NS100 Percutaneous Electrical Nerve Stimulator
An FDA-cleared, in-office neurostimulation device that reduces neuropathic pain by 87% and improves blood glucose by 37% — with zero adverse events across all published studies.
What Is Painful Diabetic Neuropathy?
Diabetic peripheral neuropathy (DPN) is a common and progressive complication of type 1 and type 2 diabetes, caused by chronic damage to peripheral nerves from prolonged elevated blood sugar levels. It affects an estimated 50% of people living with diabetes — and of those, approximately 40-50% develop painful symptoms that significantly impact daily life.
Painful diabetic neuropathy (PDN) typically presents as burning, stabbing, or shooting pain in the feet and lower legs, often worsening at night. Patients frequently experience tingling, numbness, and heightened sensitivity to touch. Beyond the physical symptoms, PDN is strongly associated with insomnia, anxiety, depression, reduced mobility, and a measurable decline in overall quality of life.
Why Standard Treatments Often Fall Short
The most commonly prescribed medications for painful diabetic neuropathy — including gabapentin (Neurontin), pregabalin (Lyrica), duloxetine (Cymbalta), tricyclic antidepressants, and opioids — provide meaningful relief for only a minority of patients. Studies consistently show that fewer than half of PDN patients achieve a 50% reduction in pain with any single medication, and most experience dose-limiting side effects including drowsiness, dizziness, weight gain, cognitive impairment, and risk of dependency.
For many patients, the reality is years of cycling through medications that provide incomplete relief while adding new side effects to manage. This is the clinical challenge that has driven growing interest in non-pharmacologic alternatives — particularly neuromodulation therapies that can address both the pain and the underlying metabolic dysfunction associated with diabetes.
The Scale of the Problem
In the United States alone, 38.4 million people are living with diabetes. The total estimated cost of diagnosed diabetes in the U.S. was $412.9 billion in 2022, including $306.6 billion in direct medical costs. The management cost of painful diabetic neuropathy can reach three times that of standard DPN care, creating significant burden for patients, providers, and the healthcare system.
For physicians managing these patients, PDN represents one of the most frustrating clinical scenarios — a large, growing population with limited effective treatment options and declining quality of life despite best efforts with available medications.
The NS100 was developed to address this unmet need — a non-pharmacologic, FDA-cleared neurostimulation device with published evidence demonstrating 87% pain reduction and 37% blood glucose improvement in patients with painful diabetic neuropathy.
87%
PAIN SCORE REDUCTION
37%
BLOOD GLUCOSE REDUCTION
NRS 7.92 → 1.04 at 90 days
80%
STOPPED ALL MEDICATIONS
HbA1c 8.9% → 5.8% at 90 days
ZERO
ADVERSE EVENTS
Pain + diabetes meds eliminated
Across all published studies

Published Clinical Evidence
Peer-reviewed and multi-center data demonstrating significant pain reduction, glycemic improvement, and medication elimination in patients with painful diabetic neuropathy.
-
Staats PS, et al. 83 patients with moderate-to-severe diabetes and painful DPN. Average follow-up: 7.85 months.
87% reduction in NRS pain scores (baseline 7.92 to 1.04 at 90 days)
37% reduction in blood glucose (HbA1c from 8.9% to 5.8%)
80% of a 45-patient subset stopped ALL pain and diabetes medications
Results sustained well beyond the 20-day wear period
DOI: 10.3389/fnins.2025.1644961
-
Madhuchander & Gurunath — Jeevak Multispeciality Hospital. Three-arm trial: standard PENS, variable-frequency sweep PENS, and sham device over 12 weeks.
Statistically significant pain improvement versus sham (p << 0.001)
80% reduction in analgesic requirements versus 7% for placebo
Significant improvement in insomnia, anxiety, and neuropathy scores
Sweep mode outperformed fixed-frequency stimulation
Zero adverse events across all study arms
-
Jones C, Erle S, Rhoades R. Device worn 20 days, average follow-up 67.2 days.
97.8% NRS pain improvement by Day 80 (7.9 to 0.18)
HbA1c reduced 25.3% (8.3 to 6.2)
Pain medications reduced by 67%
Improvement continued progressively after device removal
-
Cleveland Clinic Endocrinology & Metabolism Institute. Prospective trial for neurostimulation in moderate-to-severe painful diabetic neuropathy. Currently recruiting at Cleveland Clinic Main Campus.
The Unmet Need: Why Current Treatments Fall Short
Over 38 million Americans live with diabetes, and up to half of those with peripheral neuropathy develop painful symptoms that significantly impact quality of life. Standard pharmacologic approaches — gabapentin, pregabalin, duloxetine, tricyclic antidepressants, and opioids — frequently deliver limited relief while introducing tolerance, dependency, sedation, and cardiovascular risk.
Patients commonly report pain scores of 5 or higher despite being on multiple medications simultaneously. The annual management cost of painful diabetic neuropathy can reach three times that of standard DPN care, with total U.S. diabetes costs exceeding $412 billion in 2022.
This substantial unmet need has driven interest in non-pharmacologic interventions — particularly percutaneous electrical nerve stimulation targeting the auricular vagal and trigeminocervical pathways.

How It Works: Dual-Pathway Neurostimulation
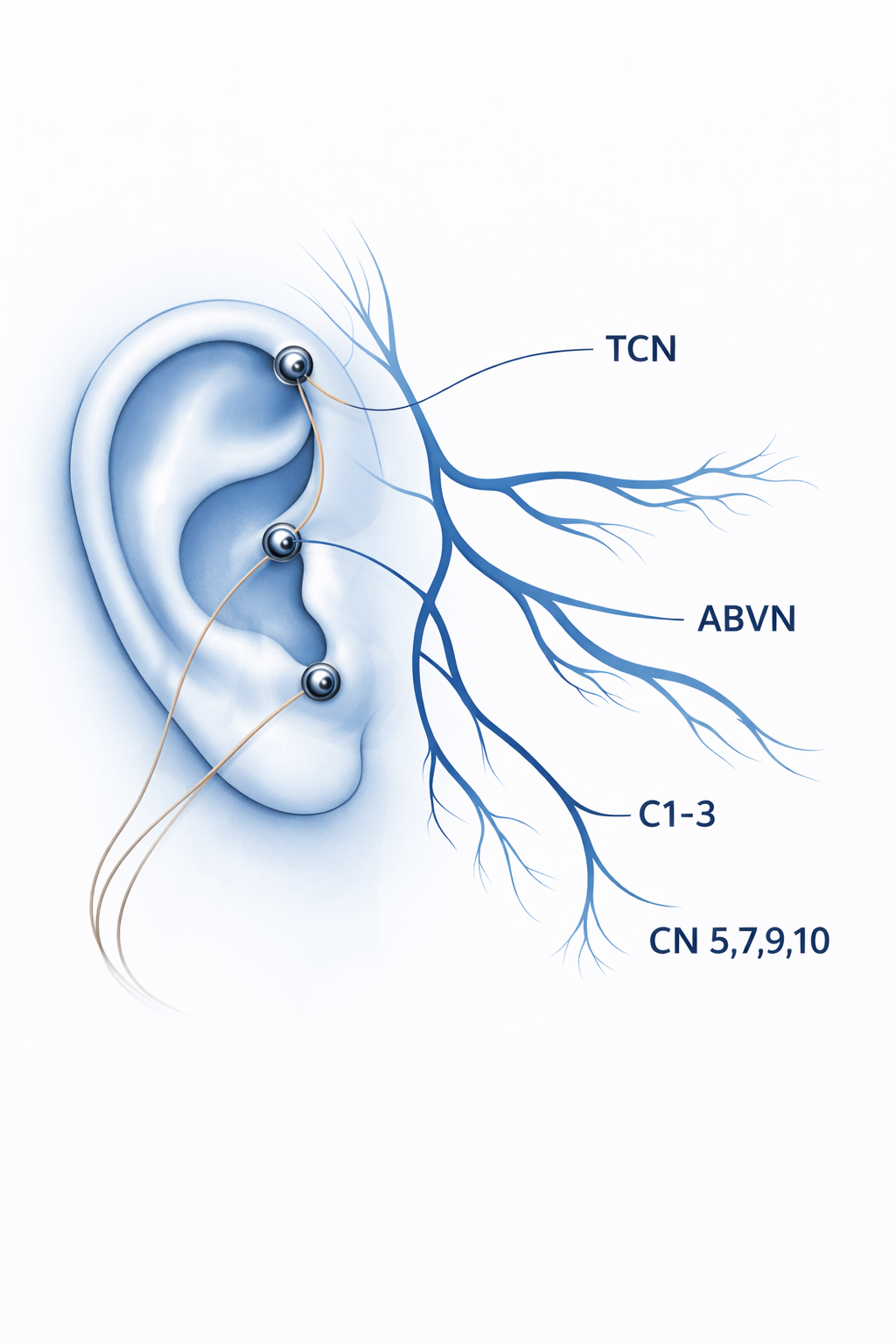
The NS100 is a wearable, battery-operated Class II medical device that delivers low-level electrical pulses through four titanium electrode arrays precisely placed in the ear. It targets two complementary neural pathways simultaneously — a dual mechanism not achievable with surface electrodes or traditional TENS.
TrigeminoCervical Pathway — Direct Pain Modulation
Electrodes targeting the auricular temporal nerve (ATN), lesser occipital nerve (LON), and greater auricular nerve (GAN) modulate C1-2-3 cervical spinal roots and cranial nerves 5, 7, 9, and 10. This provides direct pain modulation through the trigeminocervical complex via central antidromic inhibition of wide dynamic range neurons of the dorsal horn.
Vagal Pathway — Metabolic & Anti-Inflammatory Modulation
The auricular branch of the vagus nerve (ABVN) activates opioid, nicotinic, and cannabinoid receptors. This engages the cholinergic anti-inflammatory pathway, providing vagal control of metabolic state through neuroendocrine and immune response modulation — directly influencing insulin secretion and glucose regulation.
THE RESULT
This dual mechanism produces simultaneous pain reduction AND metabolic improvement. Published data suggests the NS100 may modify the underlying disease state through neuroplastic changes in pain processing and metabolic pathways — with benefits persisting well beyond the 20-day treatment period.
Treatment Process
Program & Implant
30-60 minute in-office procedure. The provider programs the NS100 via the Programmable Technical Unit (PTU) based on the patient's diagnosis. Four titanium electrode arrays are placed on auricular nerve targets using real-time impedance feedback. Can be performed by MD, DO, NP, or PA
Patient Wears Device
Worn continuously for up to 20 days. Low-level electrical pulses activate opioid, nicotinic, and cannabinoid receptors through the cholinergic anti-inflammatory pathway. Receptor-specific programming with fixed or sweep activation modes tailored to diagnosis
Remove & Follow Up
Patient returns for device removal. Over 70% report meaningful pain relief lasting up to 6 months or longer. Provider documents pain relief percentage and NRS score. The NS100 also serves as a trial for permanent neurostimulation if clinically indicated
Patient Selection Criteria
3-6 months of documented chronic pain (NRS ≥5) with diabetes diagnosis
At least one failed modality — prescription medications, physical therapy, steroid injections, OTC medications, or compression therapy
Valid ICD-10 diagnosis code: E11.42, E11.41, E10.42, or E10.41
CMS NCD 160.7.1 establishes PENS as an accepted modality
Contraindications
The NS100 is contraindicated in patients with:
Hemophilia
Pregnancy
Cognitive impairment
Implanted cardiac devices (pacemaker, defibrillator)
Indwelling or implantable stimulator devices (DBS, SCS)
For Practice Administrators: Turnkey Implementation
In partnership with NeuroDynamics Global, we manage the entire implementation process so your team can focus on patient care.
What We Handle
✓ Complete prior authorization & predetermination
✓ Full revenue cycle management (RCM)
✓ Claims billing & payer follow-up
✓ Appeals preparation & peer-to-peer support
✓ Weekly payment reconciliation reports
✓ Patient status tracking & reporting
What You Get
✓ Clinical staff training by NeuroDynamics Global
✓ All remittances post directly to your facility
✓ No upfront device purchase required
✓ Streamlined documentation process
✓ Dedicated VP of Client Development
✓ Dedicated Billing Manager assigned
Ideal for Specialty Practices:
Endocrinology • Pain Management • Neurology • Internal Medicine
Primary Care • Rheumatology • Vascular Surgery • Orthopedic Surgery
Frequently Asked Questions
-
Yes. The NS100 received 510(k) clearance (K212859) on December 20, 2021 as a Class II percutaneous electrical nerve stimulator. It was cleared by the FDA's Division of Neuromodulation and Physical Medicine Devices. Its predicate device is the Sprint PNS System (K202660).
-
Yes. The NS100 is covered by Medicare and most commercial insurers. CPT code 64555 covers the percutaneous implantation of the neurostimulator electrode array, and HCPCS code L8679 covers the implantable neurostimulator pulse generator. Redacted EOBs are available upon physician request.
-
The implantation procedure takes 30 to 60 minutes in an office or outpatient setting. It can be performed by an MD, DO, or qualified mid-level provider (NP/PA in certain states). No general anesthesia is required.
-
Over 70% of patients report meaningful pain relief lasting up to 6 months or longer. Published data shows that improvement continues progressively even after the device is removed at 20 days. In the multi-center study, patients reached near-zero pain scores (NRS 0.18) by Day 80 — well after the device was no longer active.
-
No. The NS100 is a completely non-pharmacologic treatment. In the Frontiers in Neuroscience study, 80% of a 45-patient subset stopped all pain and diabetes medications after treatment. In the RCT, analgesic requirements decreased by 80% in treatment groups compared to only 7% for placebo.
-
Unlike transcutaneous (surface) stimulation, the NS100 uses percutaneous (through-the-skin) titanium electrode arrays that target specific cranial and cervical nerve structures. This allows precise, receptor-specific neurostimulation of both pain and metabolic pathways simultaneously — an approach not achievable with surface electrodes. Published outcomes significantly exceed those reported in TENS literature for diabetic neuropathy.
-
Spinal cord stimulation requires surgery, general anesthesia, and permanent implantation. The NS100 is a minimally invasive, temporary device placed in the ear during a 30-60 minute office visit, worn for 20 days, and removed. No permanent implant, no surgery, no ongoing device management. The NS100 can also serve as a trial for permanent neurostimulation
-
The NS100 produces a dual therapeutic benefit. Through vagal nerve stimulation, the device engages the cholinergic anti-inflammatory pathway, influencing insulin secretion and glucose metabolism. In the Frontiers in Neuroscience study, patients experienced a 37% reduction in HbA1c (8.9% to 5.8%) alongside the 87% pain reduction — suggesting the device may modify the underlying metabolic disease state.
-
The NS100 can be implanted by an MD, DO, or in certain states, a qualified mid-level provider (NP or PA). NeuroDynamics Global provides clinical staff training as part of every implementation.
-
Contact us at (727) 346-8050 or info@k2wounds.com. Our team will assess your practice fit and patient volume, coordinate clinical training through NeuroDynamics Global, and handle all prior authorization, billing, and revenue cycle management.
Ready to Offer Your Patients a Proven, Non-Opioid Solution?
Contact us today to learn how the NS100 can help your diabetic neuropathy patients find lasting relief — and how our turnkey model makes implementation simple.